|
Now, molecular geometry is determined by the hybridization of the central atom. Whereas molecular geometry considers only the atoms. We know that carbon is the core atom of CS2, with two electron pairs bound (two C-S) and no lone pairs of electrons. We know that head-on overlapping is always stronger than lateral overlapping hence, they play a crucial role in determining the structure of a molecule. Molecular Geometry of Carbon Diselenide (CSe2), Hybridization in Carbon Diselenide (CSe2). As discussed earlier CS2 has a linear shape. Is our block - Study One Course At A Time a. We can easily find out the molecular geometry of any compound using the given chart. Carbon is gonna make double bonds, the both of them. In methane ammonia water and hydrogen fluoride the electron pair geometry is tetrahedral. Associate Leaving Dental Practice Letter, Ten Definition Of Education By Nigerian Scholars, Rb+1 Is Isoelectronic With Which Noble Gas, townhomes for rent in paradise valley, az. Asked Arsenic is in group five of the periodic table. Lewis, who introduced it in his 1916 article called 'The Atom and the Molecule'. The Lewis structure was named after Gilbert N. Carbon disulfide, CS2, will have a total of 16 valence electrons, 4 from the carbon atom and 6 from each of the two sulfur atoms. There's another electron pair, so it has a linear structure and that. Do not include overall ion charges or formal charges in your drawing. No tracking or performance measurement cookies were served with this page. An explanation of the molecular geometry for the IF6 + ion (Iodide hexafluoride cation) including a description of the IF6 + bond angles. The molecular geometry of sulfur dioxide is a bent shape. The volume of the gas sample increases to 9.47 L while its pressure increases to 762 mm Hg. 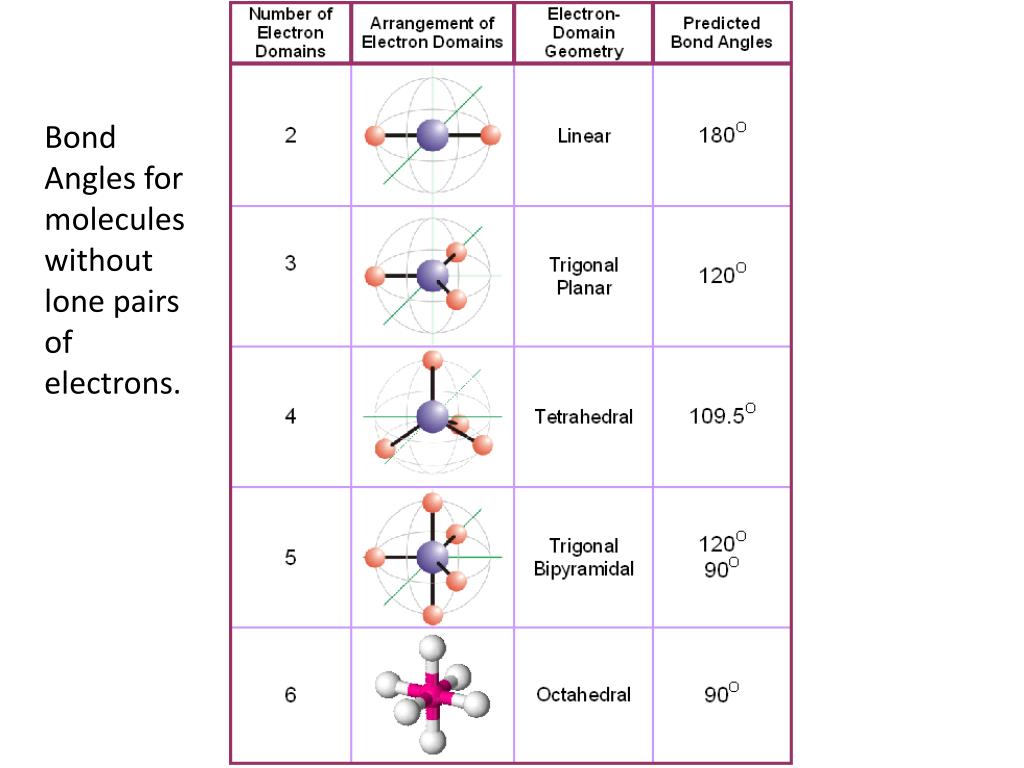
Step 2: The number of valence electrons further required to stabilize the molecule It is 8 as four are required by the carbon atom and two are required by each selenium atom. If the ideal gas law is obeyed, the temperature of the gas sample at the new volume and pressure is _ C. This makes SO2 a polar molecule like H2S. In the CS2 lewis structure, there is a total of 4 lone pairs present. The lone pairs of electrons on the central carbon atom are denoted by the letter N. It is this dipole cloud that is responsible to accept shared pair of electrons. (b) Electron pairs in molecules attract one. The electron-pair geometry around the C atom in CSe2 is There are lone pair (s) around the central atom, so the geometry of CSez is 17.  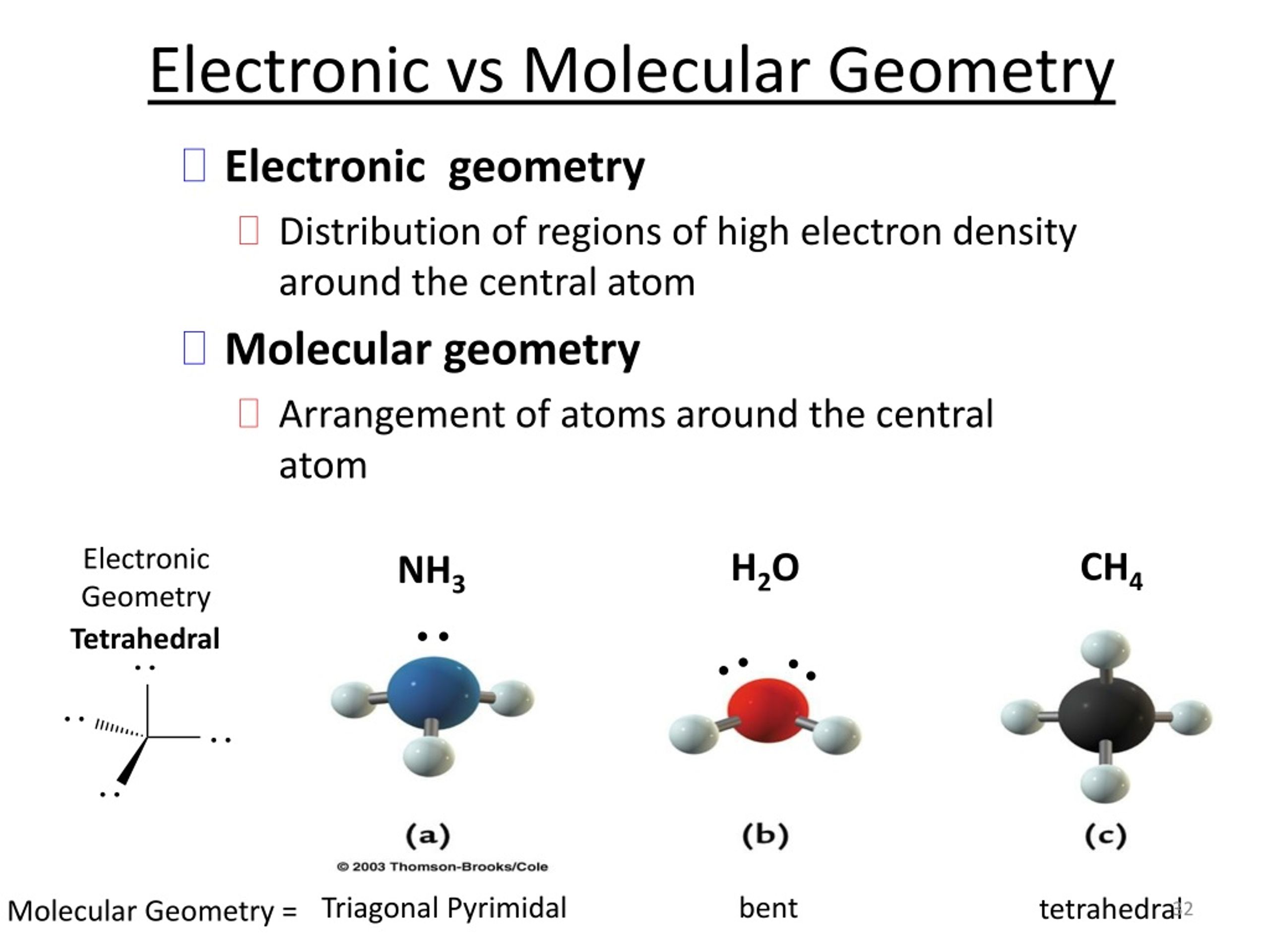
In a tetrahedral arrangement, the hybridization is s p 3 and the molecular geometry is tetrahedral. 
The double bond forming between the selenium and carbon atom has one sigma bond and one pi bond. Oxygen has a greater electronegative potential than sulfur. As I3- has one electron, this Ion has a negative charge overall. My aim is to uncover unknown scientific facts and sharing my findings with everyone who has an interest in Science. After the bond formation, it will further have 2 lone pairs and 3 BrF covalent bonds. Done on a Dell Dimension laptop computer with a Wacom digital tablet (Bamboo). These include increasing the length of the repeat unit, fusing multiple aromatic rings, altering the electron donating or withdrawing nature of the side Figure 1.3.1 (top left) The AXE formula tells you how many regions of electron density there are, which ones are specifically bonding pairs, and which ones are lone pairs. All have four pairs of electrons about the central atom (C, N, O, or F). Se=C=Se What is the name of CSe2? more covalent is the nature of the bond between them. And finally, when, H is 6, it will be Sp3d2 hybridization.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
